AVALIGN
Linear Reduction Clamp Set Instrumentation Instructions
4 Pages
Preview
Page 1
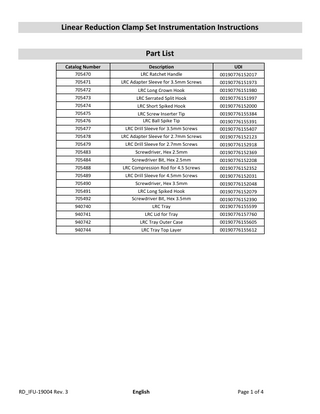
Linear Reduction Clamp Set Instrumentation Instructions Part List Catalog Number 705470 705471 705472 705473 705474
RD_IFU-19004 Rev. 3
Description LRC Ratchet Handle LRC Adapter Sleeve for 3.5mm Screws LRC Long Crown Hook LRC Serrated Split Hook
705475 705476 705477 705478 705479 705483
LRC Short Spiked Hook LRC Screw Inserter Tip LRC Ball Spike Tip LRC Drill Sleeve for 3.5mm Screws LRC Adapter Sleeve for 2.7mm Screws LRC Drill Sleeve for 2.7mm Screws Screwdriver, Hex 2.5mm
705484 705488 705489 705490 705491 705492
Screwdriver Bit, Hex 2.5mm LRC Compression Rod for 4.5 Screws LRC Drill Sleeve for 4.5mm Screws Screwdriver, Hex 3.5mm LRC Long Spiked Hook Screwdriver Bit, Hex 3.5mm
940740 940741 940742 940744
LRC Tray LRC Lid for Tray LRC Tray Outer Case LRC Tray Top Layer
English
UDI 00190776152017 00190776151973 00190776151980 00190776151997 00190776152000 00190776155384 00190776155391 00190776155407 00190776152123 00190776152918 00190776152369 00190776152208 00190776152352 00190776152031 00190776152048 00190776152079 00190776152390 00190776155599 00190776157760 00190776155605 00190776155612
Page 1 of 4
English EN Linear Reduction Clamp Set Instrumentation Instructions INTENDED USE •
The Linear Reduction Clamp Set is intended to offer a comprehensive set of surgical instruments to aide in the reduction of bone fractures during surgical reconstruction.
INTENDED USER PROFILE • •
Surgical procedures should be performed only by persons having adequate training and familiarity with surgical techniques. The physician’s education, training, and professional judgement are necessary to determine the most appropriate device and treatment option. Consult medical literature relative to techniques, complications and hazards prior to performance of any surgical procedure. Before using the product, all instructions regarding its safety features must be read carefully.
DEVICE DESCRIPTION • • • • • •
Surgical instruments comprising fixed assemblies, simple hinged instruments and simple assemblies generally composed of medical grade stainless steels, titanium, aluminum and silicone rubber. Instrument case and trays may consist of different materials including stainless steels, aluminum and silicone mats. Devices are supplied NON-STERILE and must be inspected, cleaned, lubricated and sterilized before each use. Devices require terminal sterilization per this IFU. Devices are not implantable. The following catalog instruments are manufactured and supplied by Stryker® and are compatible with the Linear Reduction Clamp Set instrumentation and/or case & tray: Catalog Number
Description
705482
Scaled Drill, 2.0x390mm
703966
Scaled Drill, 2.5x450mm
703974
Scaled Drill, 3.2x450mm
338610_-750
Small Cortical Screw Ø3.5
340614_-750
Cortical Screw Ø4.5
349608_-660
Small Cortical Screw Ø2.7
705466
Spiked Disk, Oval
705465
Spiked Disk, Large
390019
Washer Ø9mm
CONTRAINDICATIONS •
These conditions increase the risk of undesirable outcomes: o Infection o Inadequate blood supply to the operative site due to compromised vascularity. o Inadquate tissue coverage over the operative site o Bone quality compromised by disease, infection or prior surgical procedures (implanation) that cannot provide adequate support or fixation of the devices. o Foreign body (material) sensitivity o Patients with mental or neurological conditions which increase the risk of complications in post-operative care
WARNINGS • • • • • • • • • •
Avalign recommends thorough manual and automated cleaning of medical devices prior to sterilization. Automated methods alone may not adequately clean devices. Devices should be reprocessed as soon as possible following use. Instruments must be cleaned separately from cases and trays. All cleaning agent solutions should be replaced frequently before becoming heavily soiled. Prior to cleaning, sterilization and use, remove all protective caps carefully. All instruments should be inspected to ensure proper function and condition. Do not use instruments if they do not perform satisfactorily. Lubrication of quick connect related mechanisms is required prior to use to prevent difficulty in actuation. The sterilization methods described have been validated with the devices in predetermined placement locations per the case and tray designs. Areas intended for specific devices shall contain only those devices. Risk of damage – The surgical instruments are precision devices. Careful handling is important for the accurate functioning of the devices. Improper external handling can cause the devices to malfunction. Use caution when handling sharp instruments to avoid injury. Wash the instrument case and trays with an aluminum safe, neutral pH detergent to avoid faded surface colors and deterioration of anodized surfaces. If a device is/was used in a patient with, or suspected of having Creutzfeldt-Jakob Disease (CJD), the device cannot be reused and must be destroyed due to the inability to reprocess or sterilize to eliminate the risk of cross-contamination.
CAUTION Federal U.S. Law restricts this device to sale, distribution, and use, by, or on order of a physician. LIMITATIONS ON REPROCESSING Repeated processing has minimal effect on these instruments. End of life is normally determined by wear and damage due to use. DISCLAIMER It is the responsibility of the reprocessor to ensure reprocessing is performed using equipment, materials and personnel in the reprocessing facility and achieves the desired result. This requires validation and routine monitoring of the process. Any deviation by the reprocessor from the instructions provided must be properly evaluated for effectiveness and potential adverse consequences.
RD_IFU-19004 Rev. 3
English
Page 2 of 4
Reprocessing Instructions TOOLS AND ACCESSORIES
Water
Cleaning Agents
Accessories
Equipment
Cold Tap Water (< 20°C / 68°F) Warm Water (38°- 49°C / 100°- 120°F) Hot Tap Water (> 40°C / 104°F) Deionized (DI) or Reverse Osmosis (RO) Water (ambient) Neutral Enzymatic Detergent pH 6.0-8.0 i.e. MetriZyme, EndoZime, Enzol Assorted Sizes of Brushes and/or Pipe Cleaners with Nylon Bristles Sterile Syringes or equivalent Absorbent, Low Lint Disposable Cloths or equivalent Soaking Pans Medical Compressed Air Ultrasonic Cleaner (Sonicator) Automated Washer
POINT-OF-USE AND CONTAINMENT 1) 2) 3)
Follow health care facility point of use practices. Keep devices moist after use to prevent soil from drying and remove excess soil and debris from all lumens, surfaces, crevices, sliding mechanisms, hinged joints, and all other hard-to-clean design features. Suction or flush lumens with a cleaning solution immediately after use. Follow universal precautions and contain devices in closed or covered containers for transport to central supply.
MANUAL CLEANING 4) 5)
6) 7)
8) 9)
10) 11) 12)
Rinse devices under cold running tap water for a minimum of 3 minutes while wiping off residual soil or debris. Actuate moveable mechanisms and flush all lumens, cracks and/or crevices while rinsing. Prepare an enzymatic cleaning solution per manufacturer’s instructions including dilution/concentration, water quality and temperature. Immerse devices and soak for a minimum of 10 minutes. While in the solution, use a soft, bristle brush to remove all traces of blood and debris from the device, paying close attention to threads, crevices, seams, and any hard to reach areas. a. If the device has sliding mechanisms or hinged joints, actuate the device while scrubbing to remove trapped soil. b. If the device contains a lumen, use a tight-fitting nylon brush or pipe cleaner while pushing in and out with a twisting motion to facilitate removal of debris; ensure the full diameter and depth of the lumen is accessed. Flush the lumen, three times minimum, with a syringe containing a minimum solution of 60mL. Remove devices and rinse/agitate in cold tap water for a minimum of 3 minutes. Actuate moveable mechanisms and flush all lumens, cracks and/or crevices while rinsing. Prepare a neutral detergent cleaning solution per manufacturer’s instructions including dilution/concentration, water quality and temperature. Immerse devices and soak for a minimum of 5 minutes. While in the solution, use a soft, bristle brush to remove all traces of blood and debris from the device, paying close attention to threads, crevices, seams, and any hard to reach areas. a. If the device has sliding mechanisms or hinged joints, actuate the device while scrubbing to remove trapped soil. b. If the device contains a lumen, use a tight-fitting nylon brush or pipe cleaner while pushing in and out with a twisting motion to facilitate removal of debris; ensure the full diameter and depth of the lumen is accessed. Flush the lumen, three times minimum, with a syringe containing a minimum solution of 60mL. Remove devices and rinse/agitate in cold tap water for a minimum of 3 minutes. Actuate moveable mechanisms and flush all lumens, cracks and/or crevices while rinsing. Prepare an enzymatic cleaning solution using hot water per manufacturer’s recommendations in an ultrasonic unit. Sonicate the devices for a minimum of 15 minutes using a minimum frequency of 40 kHz. It is recommended to use an ultrasonic unit with flushing attachments. Devices with lumens should be flushed with cleaning solution under the surface of the solution to ensure adequate perfusion of channels. Remove devices and rinse/agitate in ambient DI/RO water for a minimum of 4 minutes. Actuate moveable mechanisms and flush all lumens, cracks and/or crevices while rinsing. Flush internal lumens a minimum of 3 times with RO/DI water (minimum of 15mL) using an appropriately sized syringe. If available, use flush ports for flushing. Dry the device using an absorbent cloth. Dry any internal areas with filtered, compressed air. Visually inspect the device for soil including all actuating mechanisms, cracks, crevices, and lumens. If not visibly clean, repeat steps 4-12.
AUTOMATED CLEANING Note: All devices must be manually pre-cleaned prior to any automated cleaning process, follow steps 1-8. Steps 9-12 are optional but advised. 13)
Transfer the devices to an automatic washer/disinfector for processing per the below minimum parameters. Phase Pre-wash 1 Enzyme Wash Wash 1 Rinse 1 Purified Water Rinse Drying
14) 15)
Time (minutes) 02:00 02:00 02:00 02:00 02:00 15:00
Temperature
Detergent Type & Concentration
Cold Tap Water Hot Tap Water 63°C / 146°F Hot Tap Water 146°F / 63°C 194°F / 90°C
N/A Enzyme Detergent Neutral Detergent N/A N/A N/A
Dry excess moisture using an absorbent cloth. Dry any internal areas with filtered, compressed air. Visually inspect the device for soil including all actuating mechanisms, cracks, crevices and lumens. If not visibly clean, repeat steps 4-8, 13-15.
DISINFECTION • •
Devices must be terminally sterilized (See § Sterilization). Avalign instruments are compatible with washer/disinfector time-temperature profiles for thermal disinfection per ISO 15883.
INSPECTION AND FUNCTIONAL TESTING • • • •
Visually inspect devices for damage or wear, including sharp edges. Instruments with broken, cracked, chipped or worn features, should not be used, but should be replaced immediately. Verify device interfaces (junctions and threads) continue to function as intended without complications. Check for smooth movement of hinges. Locking mechanisms should be free of nicks. Lubricate before autoclaving with Instra-Lube, or a steam permeable instrument lubricant.
RD_IFU-19004 Rev. 3
English
Page 3 of 4
PACKAGING • • • •
Only FDA cleared sterilization packaging materials should be used by the end user when packaging the devices. The end user should consult ANSI/AAMI ST79 or ISO 17665-1 for additional information on steam sterilization. Sterilization Wrap o Cases may be wrapped in a standard, medical grade sterilization wrap using the AAMI double wrap method or equivalent. Rigid Sterilization Container o For information regarding rigid sterilization containers, please refer to appropriate instructions for use provided by the container manufacturer or contact the manufacturer directly for guidance.
STERILIZATION Sterilize with steam. The following are minimum cycles required for steam sterilization of Avalign devices: Note: The following catalog instruments are manufactured and supplied by Stryker®. These instruments were validated within the Avalign instrument set and can be placed in the set for sterilization according to the steam sterilization parameters identified below. Description Catalog Number
1.
• • •
Scaled Drill, 2.0x390mm
703966
Scaled Drill, 2.5x450mm
703974
Scaled Drill, 3.2x450mm
US Sterilization Wraps: Cycle Type Prevacuum Prevacuum
•
705482
Temperature 132°C (270°F) 134°C (273°F)
Exposure Time 4 minutes 3 minutes
Pulses 4 4
Drying Time 30 minutes 30 minutes
The operating instructions and guidelines for maximum load configuration of the sterilizer manufacturer should be followed explicitly. The sterilizer must be properly installed, maintained, and calibrated. Time and temperature parameters required for sterilization vary according to type of sterilizer, cycle design, and packaging material. It is critical that process parameters be validated for each facility’s individual type of sterilization equipment and product load configuration. A facility may choose to use different steam sterilization cycles other than the cycle suggested if the facility has properly validated the cycle to ensure adequate steam penetration and contact with the devices for sterilization. Note: rigid sterilization containers cannot be used in gravity steam cycles. Water droplets and visible signs of moisture on sterile packaging/wrap or the tape used to secure it may compromise the sterility of the processed loads or be indicative of a sterilization process failure. Visually check outside wrap for dryness. If there are water droplets for visible moisture observed the pack or instrument tray is considered unacceptable. Repackage and re-sterilize the packages with visible signs of moisture.
STORAGE • •
After sterilization, instruments should remain in sterilization packaging and be stored in a clean, dry cabinet or storage case. Care should be taken when handling devices to avoid damaging the sterile barrier.
MAINTENANCE • •
Attention: Apply autoclavable lubricant only on the connecting elements (locking mechanism) and moving parts. Discard damaged, worn or non-functional devices.
WARRANTY • •
All products are guaranteed to be free from defects in material and workmanship at the time of shipping. Avalign instruments are reusable and meet AAMI standards for sterilization. All our products are designed and manufactured to meet the highest quality standards. We cannot accept liability for failure of products which have been modified in any way from their original design.
CONTACT Manufactured by: Avalign Technologies 8727 Clinton Park Drive Fort Wayne, IN 46825 1-877-289-1096 www.avalign.com [email protected] Distributed by: Stryker® Symbols Glossary Symbol
RD_IFU-19004 Rev. 3
Title
Symbol
Title and Translations
Manufacturer & Date of Manufacture
Caution
Lot Number / Batch Code
Non-Sterile
Catalogue Number
Federal Law (USA) restricts this device to sale by or on the order of a physician
Consult Instructions for Use
Medical Device
English
Page 4 of 4